As a regulatory T-cell intrinsic immune checkpoint, CTLA-4 is a double-edged sword: protecting us against autoimmune diseases but allowing cancer evasion of host immunity. Drugs currently in clinical use or testing showed good anti-cancer activity but cause immunotherapy-related adverse events (irAEs) in a high percentage of patients with cancer due to the reduced regulation of the immune system. Managing the delicate balance of the two functions is the major challenge for cancer immunotherapy.
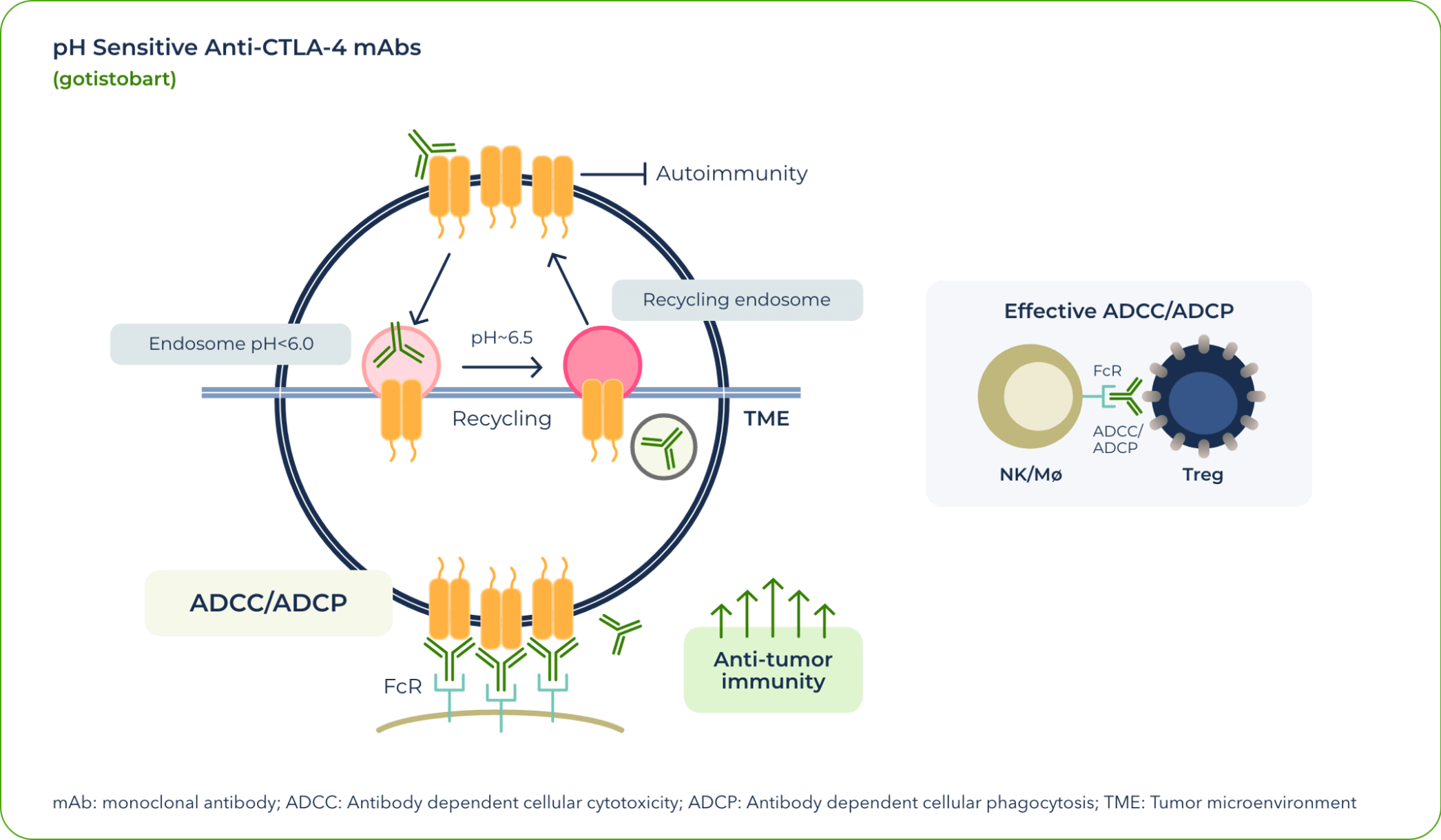
In currently approved treatments that exploit the CTLA-4 pathway, CTLA-4 degrades once dragged into lysosome in the T-cells by anti-CTLA-4 antibodies. This means that the CTLA-4 proteins are no longer available for effectively targeting regulatory T cells by anti-CTLA4 therapies, reducing clinical activities of the anti-CTLA-4 antibodies. At the same time, degradation of the CTLA-4 immune-tolerance checkpoint by the currently approved antibodies increased their toxicity.
Our team identified that the differential pH sensitivity of anti–CTLA-4 antibodies to the extracellular versus intracellular environment is critical to preserving CTLA-4 protein—a valuable insight gained during the development of gotistobart.
After gotistobart binds to the CTLA-4 receptor at the cell surface, the complex is internalized. As a result of the pH change, the mAb unbinds and allows the CTLA-4 protein to return to the surface and continue its role in anticancer signaling.
In effect, gotistobart was designed to selectively deplete the tumor-infiltrating regulatory T cells by preserving CTLA-4 recycling.1 In the peripheral tissues, preserving CTLA-4 recycling also preserves its ability to control autoreactive T cells, thus reducing IrAEs.
As a next-generation anti-CTLA-4 antibody, gotistobart is being co-developed clinically by BioNTech and OncoC4.
Gotistobart is currently being evaluated in an ongoing Phase 1/2 trial, PRESERVE-001, (NCT04140526) in patients with advanced solid tumors as a single agent or in combination with pembrolizumab, and an ongoing registrational Phase 3 trial, PRESERVE-003 (NCT05671510) evaluates the candidate as monotherapy in patients with metastatic, immunotherapy-resistant non-small cell lung cancer (NSCLC).
Gotistobart showed meaningful clinical activity in patients with solid tumors and in those with metastatic non–small cell lung cancer without driver mutations whose disease had progressed on prior lines of immunotherapy-based regimens.
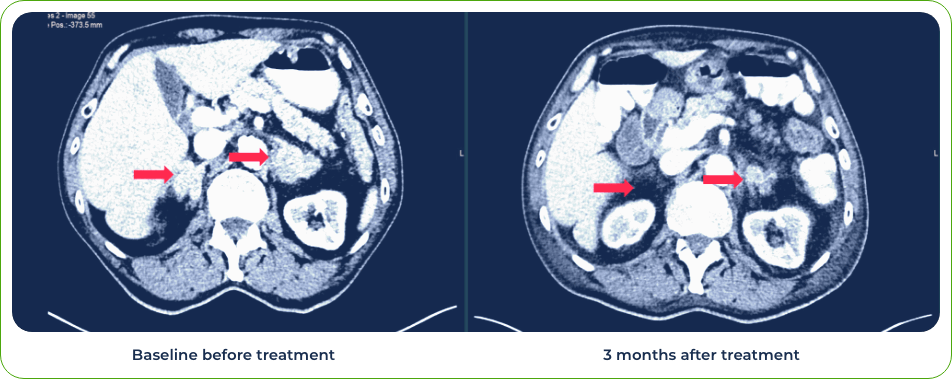
Diagnosis:
Stage IV lung adenosquamous carcinoma
Sites of metastases:
Adrenal glands and brain
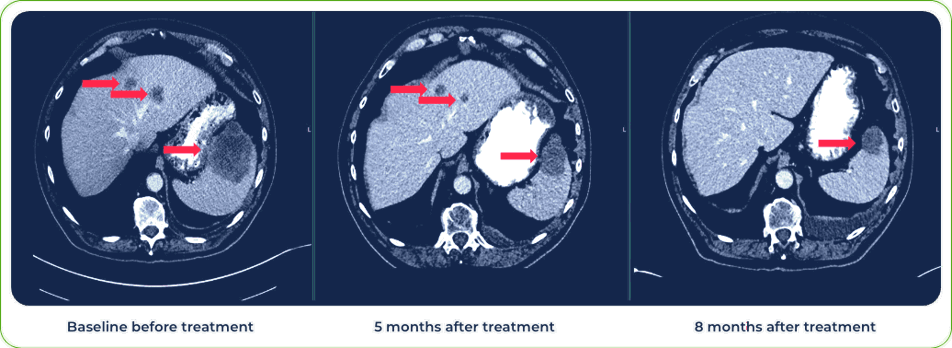
Diagnosis:
Stage IV Squamous Cell Carcinoma of Lung
Sites of metastases:
Spleen and liver
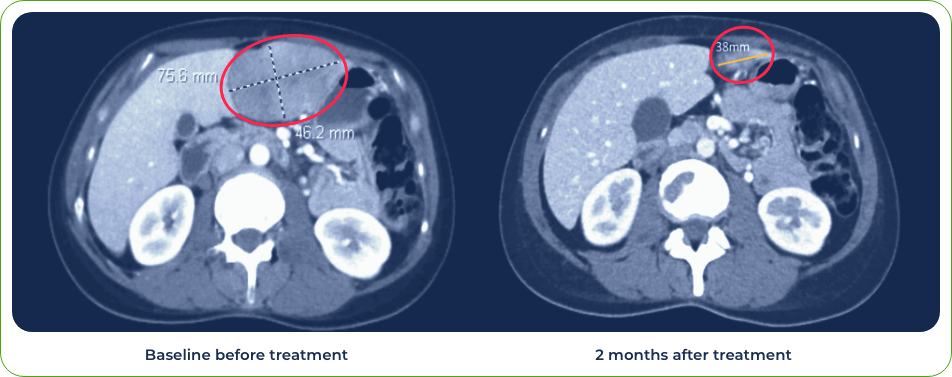
Diagnosis:
Stage IV metastatic melanoma
Single dose induced regression of large metastatic lesion in the liver

PD-1 is another protein that acts as an “off switch” for T cells, helping to regulate the immune system. PD-1 inhibitors are often given in combination with other checkpoint inhibitors.
We are currently evaluating gotistobart in a Phase 2 trial as a combination therapy with pembrolizumab in platinum-resistant ovarian cancer (NCT05446298). It is also being evaluated in the PRESERVE-006 (NCT05682443) Phase 2 trial in combination with PLUVICTO® (lutetium Lu 177 vipivotide tetraxetan) in patients with metastatic castration resistant prostate cancer who have progressed on androgen receptor pathway inhibitors and the chemotherapy taxane agents.
1. Zhang Y, Du X, Liu M, Tang F, Zhang P, Ai C, Fields JK, Sundberg EJ, Latinovic OS, Devenport M, Zheng P, Liu Y. Hijacking antibody-induced CTLA-4 lysosomal degradation for safer and more effective cancer immunotherapy. Cell Res 2019 Aug;29(8):609-627. doi: 10.1038/s41422-019-0184-1


